 
So the ionic compound formed from sodium and chlorine is sodium chlor ide. The second part of the name is based upon the anion, but has the ending ide. 
For ionic compounds such as NaCl, the first part of the name is simply that of the metal forming the cation, in this case sodium. In this case these consist of Na + cations and Cl - anions. The chemical formula of NaCl implies that there is 1 Na for each Cl. These reactions are important for maintaining the homeostasis of our blood.\) shows the best known ionic compound, sodium chloride, NaCl (common table salt). However, the carbonic acid can also leave the body as carbon dioxide gas (via exhalation) instead of being converted back to bicarbonate ion, thus driving the reaction to the right by the chemical law known as law of mass action. To return to the example of excess hydrogen ions in the blood, the formation of carbonic acid will be the major direction of the reaction. In biological reactions, however, equilibrium is rarely obtained because the concentrations of the reactants or products or both are constantly changing, often with a product of one reaction being a reactant for another. If carbonic acid were added to this system, some of it would be converted to bicarbonate and hydrogen ions. These situations of reversible reactions are often denoted by a chemical equation with a double headed arrow pointing towards both the reactants and products.įor example, in human blood, excess hydrogen ions (H +) bind to bicarbonate ions (HCO 3 –) forming an equilibrium state with carbonic acid (H 2CO 3). This back and forth continues until a certain relative balance between reactants and products occurs-a state called equilibrium. In reversible reactions, reactants are turned into products, but when the concentration of product goes beyond a certain threshold (characteristic of the particular reaction), some of these products will be converted back into reactants at this point, the designations of products and reactants are reversed. Reversible reactions are those that can go in either direction. The equations that describe these reactions contain a unidirectional arrow and are irreversible. Some chemical reactions, such as the one shown above, can proceed in one direction until the reactants are all used up. Water, salt, and sugar are examples of compounds. Explanation -A compound is a substance formed when two or more elements are chemically joined. The oxygen atoms in an O 2 molecule are joined by a double bond. A molecule is, in general, a group of two or more atoms that are chemically bonded together, that is, tightly held together by attractive forces. The atoms are bonded together, and the compound has different physical and chemical properties than the individual elements. 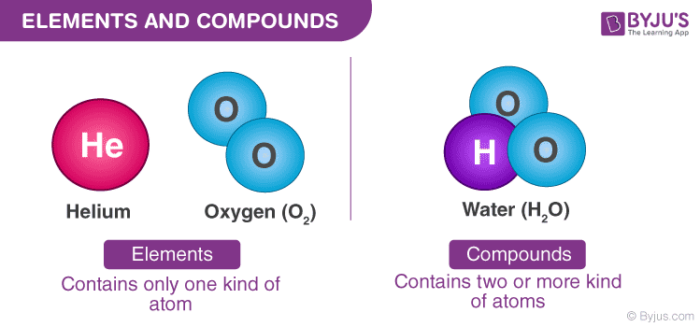
Molecular oxygen, on the other hand, as shown in Figure 2, consists of two doubly bonded oxygen atoms and is not classified as a compound but as an element.įigure 2. According to the law of conservation of matter, the number of atoms before and after a chemical reaction should be equal, such that no atoms are, under normal circumstances, created or destroyed.ĢH 2O 2 (hydrogen peroxide) → 2H 2O(water) + O 2 (oxygen)Įven though all of the reactants and products of this reaction are molecules (each atom remains bonded to at least one other atom), in this reaction only hydrogen peroxide and water are representative of a subclass of molecules known as compounds: they contain atoms of more than one type of element. This is an example of a balanced chemical equation, wherein the number of atoms of each element is the same on each side of the equation. In the equation below, the reaction includes two hydrogen peroxide molecules and two water molecules. The reactant hydrogen peroxide is broken down into water, containing one oxygen atom bound to two hydrogen atoms (H 2O), and oxygen, which consists of two bonded oxygen atoms (O 2). An arrow is typically drawn between the reactants and products to indicate the direction of the chemical reaction this direction is not always a “one-way street.” For the creation of the water molecule shown above, the chemical equation would be:Īn example of a simple chemical reaction is the breaking down of hydrogen peroxide molecules, each of which consists of two hydrogen atoms bonded to two oxygen atoms (H 2O 2). The substances used in the beginning of a chemical reaction are called the reactants (usually found on the left side of a chemical equation), and the substances found at the end of the reaction are known as the products (usually found on the right side of a chemical equation). When two hydrogens and an oxygen share electrons via covalent bonds, a water molecule is formed.Ĭhemical reactions occur when two or more atoms bond together to form molecules or when bonded atoms are broken apart. 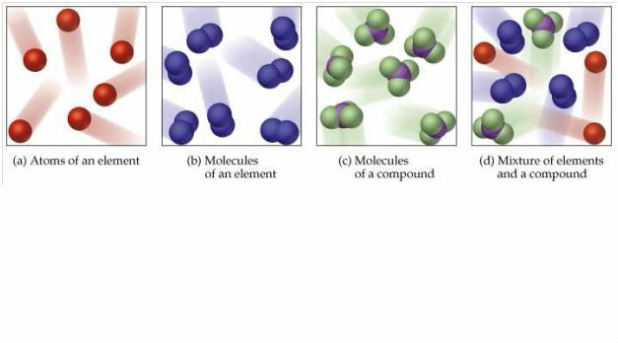
Two or more atoms may bond with each other to form a molecule. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
